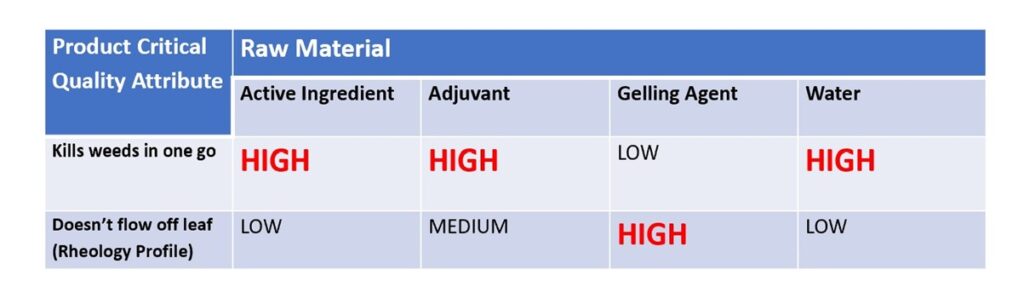
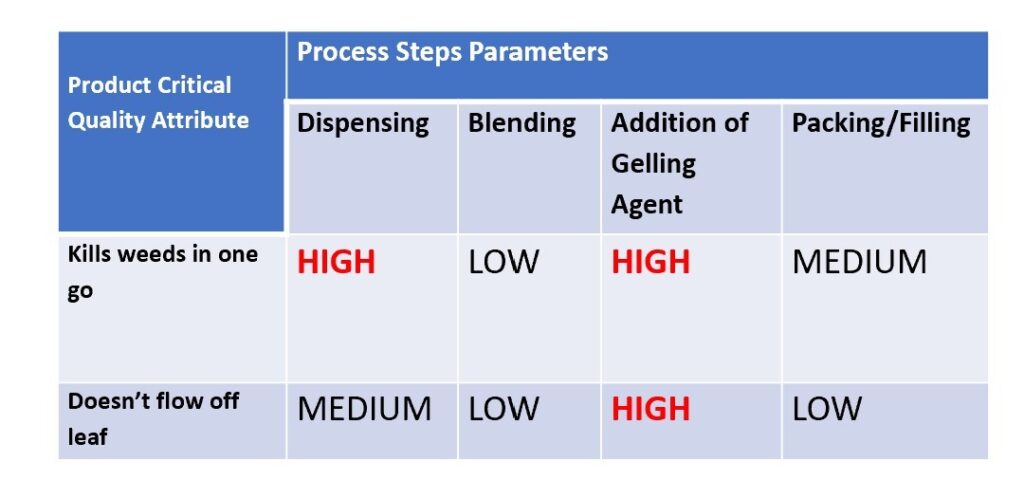
For the thrill seekers among you the sinking feeling that you experience on a roller coaster – when your stomach is suddenly very light due to less force being exerted on it – is a pleasurable one. For a formulator of suspensions or emulsions though, that “sinking feeling” is not a good one as you come in to the lab one morning to find that your particles have well and truly sunk. The option of “just shake them up” is often unacceptable to your customers, so what do you do?
For the thrill seekers among you the sinking feeling that you experience on a roller coaster – when your stomach is suddenly very light due to less force being exerted on it – is a pleasurable one. For a formulator of suspensions or emulsions though, that “sinking feeling” is not a good one as you come in to the lab one morning to find that your particles have well and truly sunk. The option of “just shake them up” is often unacceptable to your customers, so what do you do?
Our new on-line Suspension and Emulsion – ology course will provide you with many of the answers to your problems and will also give you some of the theoretical background to understand what is happening in your formulation.
One of the pioneers in the theory that describes suspensions and emulsions was Sir George Gabriel Stokes (1819-1903) who made numerous contributions to the advancement of science in fluid dynamics, optics and mathematical physics. His well known “Stokes’ Law” gave the first insight into the factors influencing the stability of solids in fluids and the principles still apply today. His law states that the velocity of sedimentation increases proportionally with the square of the particle size – i.e larger particles sink more quickly than smaller ones – and also that particles will sink more quickly in lower viscosity media. Despite some restrictions since his work was first published, these findings are still valid today. As well as helping formulators with their products, Stokes’ law also explains why small water droplets can remain suspended in air (as clouds) until they reach a critical size and start falling as rain – or as snow and hail of course!
It is not well known that Stokes was, like all scientists, a true romantic. In a letter to his fiancée Stokes spoke of his plans for the honeymoon and wrote”:
“If we are married at the time we are at present thinking of, and go to Switzerland as we talked of, I think I will bring a couple of quartz prisms, a quartz lens, and a piece of uranium glass with me, to observe the spectrum on top of the Rigi or Faulhorn!”
The good news is that you don’t need to bring any prisms along to the training course, but you can bring along your problems as there will be time on the second day to use these in a case study section
For full details of Suspension and Emulsion – ology and how to register, see the course web page.
David Calvert, March 2025